CED (Convection Enhanced Delivery) in DIPG
Diffuse intrinsic pontine glioma (DIPG) is locally highly infiltrative. In this type of tumor, cancer tissue cannot be distinguished from normal brain tissue macroscopically. This infiltrative nature makes effective therapy extremely difficult, if not impossible. To be clinically useful, a therapy must have the ability to selectively target and kill tumor cells without significant damage to the normal brain tissue. Given the need for cell specificity, surgical resection and stereotactic radiosurgery have limited utility, pose a great risk of injury and are therefore rarely an option to consider.
Conventional radiation therapy is currently employed routinely as a palliative approach. The improvement of newer chemotherapeutic agents’ tumor selectivity and the development of targeted therapeutic agents in recent decades raise hopes that improved chemotherapy will lead to improved outcome. Paralleling this development has been the promising advance in drug delivery to the central nervous system via local delivery systems to overcome the blood-brain barrier (BBB), one of the major hurdles in delivering drugs to the brain.
CED vs Intra-Arterial
Intra-arterial is an intravascular route of delivery that is regional. A small catheter is inserted into an artery of the brain at which time chemotherapy is injected. The chemotherapy does get into the systemic circulation and ultimately the rest of the body.
CED, or convection enhanced delivery, injects agents directly into the tumor. It relies on placing a small catheter in the brain tumor tissue and using a slow rate of infusion. This is the most immediate method for achieving very high concentrations of drug into the tissue WITHOUT any appreciable systemic exposure. It essentially eliminates the potential for systemic toxicity (i.e. bone marrow suppression, sepsis, renal or hepatic dysfunction, etc.).
We will be discussing the application of convection-enhanced delivery (CED), also known as interstitial infusion, in the treatment of DIPG. CED is a technique designed to deliver drugs directly into the tumor at high concentrations. This avoids or at least greatly reduces systemic exposure to the drug. Drugs being studied for delivery through CED include conventional chemotherapy drugs, novel small molecule agents and macromolecules such as therapeutic antibodies, immunotoxins, and viral vectors, some of which would otherwise never gain access to the brain.
Blood-Brain Barrier
Chemotherapy may be administered systemically or locally. In systemic chemotherapy, the drug is administered orally or intravenously. An important limitation of systemic chemotherapy in the treatment of brain tumors is the existence of the blood-brain barrier (BBB). The BBB isolates the circulating blood from cerebrospinal fluid (CSF) in the central nervous system (CNS). It occurs along cerebral capillaries and consists of tight junctions (zona occludens) that do not exist in systemic circulation. Endothelial cells joined by tight junctions restrict the entry of microscopic objects (e.g., bacteria) and large or hydrophilic molecules into the CSF, while allowing the diffusion of small hydrophobic molecules (O2, certain hormones, CO2, etc.). Typically, molecules with molecular weight greater than approximately 40kD are unlikely to penetrate the intact barrier. For the brain’s supply of nutrients and removal of metabolites, cells of the barrier actively transport substances such as glucose across the barrier with specific proteins (transporters). The BBB acts effectively to protect the brain from many common bacterial infections and some toxic substances. Yet it presents a major challenge in delivering therapeutic agents to specific regions of the brain for the treatment of brain tumors and other disorders. Most cancer drugs are not able to permeate the BBB because they are polar in structure or too large in molecular weight. Even for drugs that are able to cross the cerebral capillary bed, it is difficult to achieve optimal concentrations due to systemic toxicity.
Another difficulty in the delivery of drugs for the treatment of brain tumors is how to direct those agents to the specific anatomic region or tumor mass to reduce the disturbance of normal neurological functions. Several strategies have been developed in an attempt to overcome this barrier, including: 1) the temporary disruption of the BBB, 2) modification of drugs to enhance their ability to permeate the BBB and 3) local delivery methods such as intratumoral/intra-cavitary embedding of drug-containing polymers or microchips, intra-arterial injection, direct injection of drugs into the tissue or CSF in the ventricles or subarachnoid space, and CED to deliver drugs directly into the extracellular space.
Local Delivery
Direct injection into the tumor or CSF is one of the earliest local delivery methods attempted. When injected into the tumor, it relies on diffusion for the drug to reach the cancer cells not directly adjacent to the injection site. Therefore the drug has an uneven distribution and can only reach the tumor tissue that is a short distance from the injection site. With small molecules, depth of distribution is often limited to several millimeters, with an exponential decay in concentration from the point source. Thus, the distribution of therapeutic concentrations of a drug is limited to a small volume of tissue around the injection site, often with very high and sometimes toxic concentration at the center. Drugs can also be injected directly into the CSF, and the drug is usually only able to reach a shallow layer of the brain using this technique.
Drug-containing polymers and microchips are a more recent development and they can be embedded at the time of surgical resection of brain tumors. As in the case of direct injection, this method relies on diffusion for the drug to spread past the embedding site and has similar limited and uneven distribution.
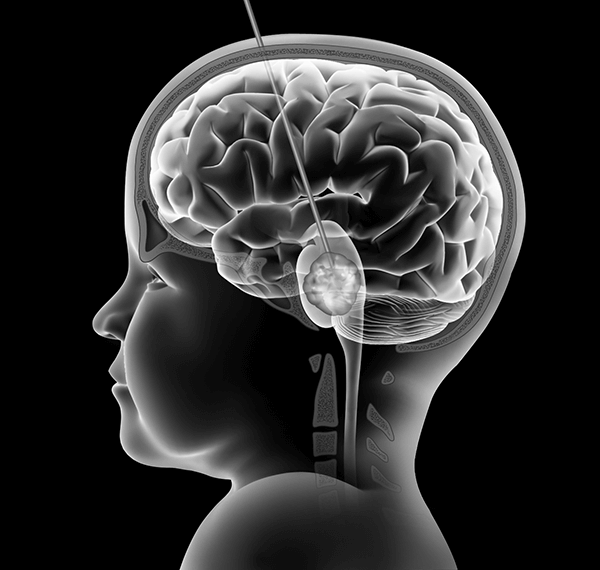
Convection-Enhanced Delivery
Convection-enhanced delivery (CED) is a novel drug delivery method first developed by a research group directed by Edward Oldfield at the National Institute of Neurological Disorders (NINDS) in the early 1990s. This method was named convection-enhanced delivery because the therapeutic molecules are distributed into the extracellular space driven by a small, persistent hydrostatic pressure generated by an infusion pump, essentially, forced convection of a fluid containing a therapeutic agent. In contrast to diffusion which depends on a concentration gradient to distribute the molecules, the use of hydrostatic pressure in CED allows for the distribution of a homogeneous concentration of small and large molecules over large distances by displacing extracellular fluid with the infusate (fluid infused). In practice, the agent is delivered into the parenchyma or tumor through a microcatheter, or multiple microcatheters, inserted into the tissue. Infusion rates typically range from 0.1-10µl/min. The distribution from a single point source results in an elliptical to spherical distribution and spatial distribution is in some degree dependent on the tissue type (i.e., grey matter versus white matter). In a given tissue type, distribution volume is approximately linear to infusion volume.
CED into brain parenchyma, both white and gray matter, has shown reproducible large volumes of distribution with homogeneous drug concentration. Oldfield group’s initial work showed that the concentration fall-off at the border is steep, resulting in a potentially large benefit in the delivery of cancer drugs in reducing toxicity to surrounding normal brain tissue.
Several factors influence the distribution volume. One key factor to achieve a large volume of distribution is the stability of the agent in the extracellular space. Lipophilic agents may be exported transvascularly through blood vessels leading to a high efflux of the drug and limited distribution. Some other drugs may be prone to enzymatic degradation in the extracellular space. Another important determinant for distribution of macromolecules is the surface characteristics of the molecule and the extracellular matrix, i.e., the substances in the extracellular space within tissues that serve various purposes, including but not limited to serving as scaffolding to hold tissues together and helping to determine the behaviors of the cells. Binding of the molecule to the extracellular matrix or surface receptors may limit distribution. Binding to cell surface receptors may be overcome by saturating receptors with excess ligands. Binding of macromolecules to extracellular matrix has been overcome with some success by co-infusion of heparin.
Size of the molecules also affects volume of distribution. Early CED studies by Oldfield group and others suggested that 180kD, the size of immunoglobulin G (IgG), appeared to be the largest size that could pass through the extracellular space without the need of surface modification to the extracellular matrix. Recently, with the help of surface modification, adeno-associated virus (AAV, 40nm) and liposomes (50-200nm) have been distributed to large volumes of brain tissue. Surface modifications used were pegylation with liposomes and heparin co-infusion to saturate heparin sulfate proteoglycan (HSP) binding with AAV.
The volume of distribution is also affected by the retrograde movement of fluid along the outside of the catheter (backflow or reflux). Reflux is determined by catheter diameter, infusion rate, and tissue density among other factors. The larger the diameter of the catheter, the greater is the backflow along its outer wall. If reflux reaches a low pressure zone (necrosis or CSF space), the fluid will inadvertently be lost into these spaces. This leads to the accumulation of the drug in these regions which may cause toxicity. Finally, increasing the infusion rate can increase the overall volume of distribution; however, this may also increase backflow, potentially shunting fluid away from the target region.
Ideally, agents delivered via CED should be contained within the target region of brain parenchyma or tumor mass. However, there are low pressure regions in some tumors along which infusate will flow, sometimes into ventricles or subarachnoid space. This phenomenon is usually referred to as leakage and has often been observed in both humans and experimental animals. One study indicates that this can happen in 20% of CED procedures. This obvious waste of therapeutic agents will consequently reduce the volume of distribution and drug concentration in the planned target region. It may also cause untoward effects on normal brain tissue. It is therefore critical to follow the flow of the infused agents. When this happens, it might be helpful to adjust the placement of the catheter to move the opening away from the low pressure region. It is also not known yet whether this leakage is reversible. If reversible, pausing infusion for a period of time and subsequently restarting the infusion could eliminate leakage.
Although the physical parameters influencing drug distribution by CED have not been thoroughly clarified, the ability of CED to achieve high concentrations of a therapeutic agent over large volumes of brain tissue has led to several clinical trials in patients with neurodegenerative disorders and malignant gliomas. Therapeutic studies for malignant gliomas have focused on delivering targeted macromolecules (monoclonal antibodies, recombinant toxins, etc.) or currently available small molecule drugs.
Catheter Design for CED
Metal needles have been used as the infusion tool since the early studies of CED in laboratory animals. Most of the recent clinical trials of CED in the treatment of malignant gliomas have used ventricular catheters made of Silastic® rubber. Ideally, a catheter for CED should be reflux-free; does not adsorb therapeutic agents to its wall, especially when expensive novel targeted agents are used; and should have tip configurations that direct the drug to desired regions. In certain instances, it may be required to confirm catheter placement before drug infusion with magnetic resonance imaging (MRI) where MRI-compatible catheters are needed.
As briefly discussed above, reflux negates the bulk flow of infusate in the extracellular space that is produced by CED. In the presence of reflux, an increase in infusion volume does not produce an increase in distribution volume accordingly. Reflux causes the drug to flow into ventricular or subarachnoid space where it may cause toxicity. While reduction in infusion rate may reduce the chance of reflux, it would be ideal to have the option of infusing at various flow rates, i.e., up to 10µl/minute or more if possible, to achieve desired volume of distribution in a reasonable period of time.
Simple infusion tools such as metal needles have high rates of reflux. Several groups, including Souweidane group at Weill Cornell Medical College, observed that a step-design cannula significantly reduces, or even effectively prevents, backflow. The group used a 22 gauge guide cannula with a 28 gauge internal cannula, both of fused silica. The internal cannula extended beyond the end of the guide cannula by 5 mm. The cannula set was left in place for 5 minutes before infusion started. At flow rates as high as 8µl/minute of an 124I-labeled monoclonal antibody, no reflux was observed on positron emission tomography (PET) imaging. Presumably the tissue surrounding the extended internal cannula sealed off the entry tract. There might be a threshold that this design can withstand the pressure, but infusion rates higher than 8µl/min have not been attempted with this tip configuration. Nevertheless this design offers an attractive improvement over the cannula design previously used. This fused silica cannula set is not and probably will never be approved for clinical use due to its insufficient mechanical strength. Ventricular catheters currently used in CED clinical trials have larger diameters and would produce a higher chance of reflux based on laboratory observation of the relationship between reflux and cannula diameters. Advances in biocompatible materials such as polymers and ceramic may eventually make small diameter MRI-compatible step-design cannulas and catheters strong enough for clinical use.
Tip Configuration
A standard cannula only has an opening at its tip. In certain instances, such as after radiation therapy where scars may form inside the tumor, this may not allow for sufficient flow of infusate. Considering infusates will follow the path of least resistance, a multi-tipped cannula may provide better pressure output, and therefore, achieve a better volume of distribution. The effectiveness of the multi-opening configuration has been questioned by studies showing that a multi-port catheter delivered most of the infusate through the proximal port and thus behaved like catheters with only one port.
One research group constructed a 3-mm long porous hollow fiber catheter to increase the surface area of the brain in immediate contact with the drug releasing area. The hollow fiber has innumerous pores of 0.45µm along its walls. This theoretically avoids clogging, which happens in certain instances. The hollow fiber catheter offers up to a threefold increase in the distribution volume of the drug into the normal mouse brain when compared to a needle which has a single macroscopic pore. The tiny microscopic pores do not have the same pressure-shunting properties as the macroscopic pores do; therefore a long length of the porous wall is effective in delivering drugs. In large animal and human applications, it is more reasonable to have this porous hollow fiber configuration at the tip for a few millimeters to a few centimeters rather than the entire catheter being porous. The porous wall and step design could even be combined to reduce reflux during drug administration.
In certain other instances, it may be desirable to direct the infusate preferentially in a specific direction. Due to the pressure-shunting properties of the proximal port on the regular multi-port cannulas, it may not be effective to direct infusate distribution via such a tip configuration. One potential design is to construct a catheter with independent cannulas inside. Each cannula has an opening at a predetermined location and direction with its pressure being independently controlled. This design will require additional engineering and testing to determine its feasibility.
Monitoring Drug Distribution
Monitoring the distribution and concentration of an infused drug is critical for numerous reasons. In order for the delivered therapeutic agent to be effective, in addition to its biological effectiveness, it must be distributed within the tumor in therapeutic concentrations. Exposure of normal tissue to the drug should be controlled to reduce the probability of toxicity. It is also highly desirable to monitor for possible backflow and leakage so that cannula placement can be adjusted to correct for any problems that may arise. The importance of monitoring in vivo distribution and concentration is highlighted by the difficulty in achieving optimal therapeutic efficacy in recent clinical trials. In the recent TGFa-PE38 study and the phase III PRECISE trial for glioblastoma (see below), poor drug distribution was cited as one of the reasons for the unsatisfactory efficacy results. Monitoring the distribution and concentration of CED infusate in humans is difficult due to the fact that the majority of therapeutic agents cannot be seen on any of the clinical imaging methods. Nevertheless, distribution can be visualized under certain circumstances. T2-weighted magnetic resonance (MR) images are helpful in identifying infusate distribution in regions of relatively normal intensity, but distribution cannot be identified with certainty when infused into already hyperintense regions, such as in the case of DIPG.
Another choice is to use visible surrogate tracers. Gd-DTPA and 123I-albumin have been co-infused as surrogate tracers, viewable on T1-weighted MR and single photon emission computed tomography (SPECT) images, respectively, in clinical studies. The shortcomings of surrogate markers are that they are only able to track the initial distribution accurately. Differences in biological activities and clearance can confound their ability to follow the volumetric distribution of the therapeutic agent over time. Moreover, neither T2 MRI signals nor surrogate tracers are able to provide information on the concentration of the infused therapeutic agent. The ideal scenario is to directly image the therapeutic compound. With calibration, the concentration of the drug can be determined as well as the distribution. Utilizing serial imaging, clearance can be followed over time. In an ongoing clinical trial at Memorial Sloan-Kettering Cancer Center and Weill Cornell Medical College, a therapeutic monoclonal antibody is labeled with 124I to treat DIPG. 124I is a positron emitter that can be visualized using PET imaging at a high resolution. The spatial resolution of 124I PET is significantly higher than that of 123I SPECT. 124I has an intrinsic spatial resolution loss of only 2.3 mm. It is expected that much more detailed information regarding the distribution and concentration of CED infusate will be acquired. This approach of labeling a therapeutic agent with imageable radionuclide can be applied to some other agents and applications. For some other therapeutic agents, novel tags such as paramagnetic particles may prove useful in labeling the drug for quantitative in vivo imaging.
Predicting and Planning CED Distribution
It is critical to define the relationship between the volume of infusion (Vi) and the volume of distribution (Vd) to understand the expected distribution of an agent delivered into the brain via CED. This relationship is approximately linear and has variable slopes depending on the anatomical site of administration as well as the therapeutic compound. For instance, the Vd/Vi ratio is 8.2 in the non-human primate (NHP) striatum compared to a ratio of 4.1 in cerebral white matter for small molecules. A ratio of 8.7 was observed in the NHP brainstem for Gd-albumin (72kD). This ratio can serve as an estimate to match tumor volume in clinical trials.
BrainLAB AG (Feldkirchen, Germany) has developed a software package called iPlan Flow specifically for use in planning CED. The software takes data obtained via MRI regarding brain tissue characteristics of individual patients as input. Then the software helps in determining cannula placement, calculating the infusion parameters and predicting distribution. The plan for treatment can be visualized in three dimensions, including the number and position of catheters. One study retrospectively tested the ability of this software using MR diffusion tensor imaging to predict patient-specific drug distributions by CED. 123I-labeled albumin was co-infused as a surrogate tracer with the targeted recombinant cytotoxin IL13-PE38QQR in patients with recurrent malignant gliomas. The spatial distribution of 123I-albumin was then compared with a drug distribution simulation provided by iPlan Flow. The algorithm had a high sensitivity and specificity in identifying catheter trajectories that resulted in reflux or leakage. The mean concordance of the volume of distribution between the actual 123I-albumin distribution and the simulation was 66% and the mean maximal inplane deviation was less than 8.5 mm. The use of this simulation algorithm was considered clinically useful in 85% of the catheters. Even though albumin does not have a specific affinity towards malignant tissue compared to targeted agents, this simulation showed that software with the ability to take into account characteristics of an individual patient’s anatomy and pathophysiology is helpful in the planning of CED. iPlan Flow has yet to be tested in CED in the brainstem.
Safety of CED in the Brainstem
The concept of using CED for DIPG is appealing given that this particular tumor is relatively compact, has growth patterns simulating white matter tracts, seldom metastasizes before local relapse and has no definitive therapy. The Souweidane group first established the feasibility of this delivery route in the brainstem in small animals for potential clinical application in 2002. Subsequently, the safeties of inert agents, characteristics of distribution and toxicity of potential therapeutic agents in the brainstem of small animals and non-human primates have been studied. This approach has also been used safely in a small number of patients with brainstem diseases. These studies showed that CED does not cause clinically relevant mechanical injury to the brainstem and this approach has a promising therapeutic application in humans. In clinical practice, image-guided frameless stereotaxy can be utilized to target the brainstem in children for biopsy or cannula insertion with high accuracy and low risks of temporary or permanent morbidity. These will help establish CED as an accepted drug delivery method in the treatment of DIPG.
Therapeutic Efficacy of CED
CED of chemotherapeutic molecules has shown considerable promise in phase I and phase II clinical trials in patients with recurrent malignant gliomas. However, phase III results are less encouraging. CED in the treatment of DIPG has produced encouraging results in preclinical studies. A few phase I trials of CED in DIPG are recruiting patients or in the planning stage.
Several factors that are critical in achieving good therapeutic efficacy require further elucidation. The convective force used in CED facilitates drug distribution to larger volumes of brain tissue. However, malignant gliomas may contain areas of fibrosis and necrosis, especially after receiving external beam radiation therapy, which is currently part of the standard of care. CED, as an investigational therapy, usually is not started until the completion of radiation therapy. The fibrosis and necrosis may cause chaotic pressure gradients within the tumor and therefore an unpredictable distribution of the drug. Even within the peritumoral margins, targeting infiltrating tumor cells may be limited by the normal anisotropy of the brain tissue resulting in preferential flow of fluid away from the intended target. Furthermore, the presence of areas of disrupted BBB either by the pathological changes or by previous treatment such as radiation therapy may increase efflux of drugs out of the CNS. A better understanding of drug distribution will become a critical part of evaluating future studies employing CED. Another concern is that the drugs infused in a single session may maintain their therapeutic concentration for a period too short to be effective before being cleared out of the target region. Once we have a better understanding of drug distribution and clearance, other unsolved questions including optimal catheter design and placement, infusion rate and duration, and the benefit of repeat infusions can be better addressed.
The use of targeted macromolecules allows for either intratumoral or peritumoral treatment in malignant gliomas. Some of these agents may not be specific enough, potentially leading to injury to normal tissue. This was seen with IL4-PE, which initially started at a concentration of 2µg/ml. The potential benefit of targeting multiple molecules by combining different recombinant toxins, or combining these agents with other chemotherapies, remains unknown. Despite these limitations and uncertainties, significant responses have been observed in all of the CED clinical trials described below.
CED Clinical Trials for the Treatment of Brain Tumors
Theoretically, any antineoplastic agent can be delivered through CED for the treatment of brain tumors, including standard chemotherapeutic agents and novel macromolecules such as monoclonal antibodies and viral vectors. One unresolved issue is that CED, in its current form, is a surgery and typically performed as a single session. It is unknown how long the infused drugs remain at therapeutic concentrations after a single session of CED. Imaginatively, it is more like a bolus dose, and predictably only a portion of the cancer cells are killed by such a bolus dose and the remaining cancer cells will continue to grow, ultimately resulting in failure of treatment.
For various reasons, most standard chemotherapeutic agents do not cross the BBB in sufficient amounts to have a significant effect on the cancer. CED of such small molecules showed that these agents have observable antitumor responses. However, more neurological complications have been observed when these agents were delivered via CED compared to systemic chemotherapy. There are efforts to improve formulations of these agents for local delivery to reduce neurotoxicity and enhance therapeutic response. These efforts, if successful, will make CED of small chemotherapeutic molecules applicable on a larger scale.
Future Directions
CED of therapeutic agents in the treatment of malignant brain tumors has shown considerable promise in preclinical and some clinical studies. Future advances will occur on two fronts:
- the development of more effective therapeutic agents for delivery via CED and
- the improvement of the technique of CED.
As discussed above, the current single session CED is more like delivering a bolus dose. Clinically feasible methods to deliver multiple cycles of CED or continuous CED lasting up to several weeks are desired. This will require the development and engineering of catheters suitable for these purposes and desirably also pumps that can be embedded and allow patients to remain ambulatory.
CED-based therapies will continue to evolve, with a need for additional preclinical and clinical research.